Abstract
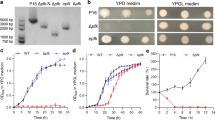
Streptomyces is studied intensively for its outstanding ability to produce bioactive secondary metabolites and for its complicated morphological differentiation process. A classical genetic manipulation system for Streptomyces has been developed and widely used in the community for a long time, using antibiotic resistance markers to select for double-crossover mutants. The screening process is always laborious and time-consuming. However, the lack of a suitable chromogenic reporter for Streptomyces has limited the use of color-based screening system to simplify the selection process for double-crossover mutants. In this study, a blue reporter system for Streptomyces has been established by mining an indigoidine synthetase gene (idgS) from Streptomyces lavendulae CGMCC 4.1386, leading to the development of a time-saving gene inactivation system for Streptomyces by simple blue-white screening. A series of Streptomyces suicide and temperature-sensitive plasmids containing the idgS reporter cassette were constructed and used successfully to inactivate genes in Streptomyces, allowing a simple and efficient screening method to differentiate the colonies for double-crossover (white) and single-crossover (blue) mutants. Inactivation of the putative γ-butyrolactone synthase gene afsA-y via the idgS-based blue-white screening method revealed that the paulomycin production is negatively controlled by afsA-y in Streptomyces sp. YN86.




Similar content being viewed by others
References
Bérdy J (2005) Bioactive microbial metabolites. J Antibiot 58:1–26
Bierman M, Logan R, O’brien K, Seno E, Nagaraja RR, Schoner B (1992) Plasmid cloning vectors for the conjugal transfer of DNA from Escherichia coli to Streptomyces spp. Gene 116:43–49
Chater KF (1993) Genetics of differentiation in Streptomyces. Annu Rev Microbiol 47:685–713
Fang J, Zhang Y, Huang L, Jia X, Zhang Q, Zhang X, Tang G, Liu W (2008) Cloning and characterization of the tetrocarcin a gene cluster from Micromonospora chalcea NRRL 11289 reveals a highly conserved strategy for tetronate biosynthesis in spirotetronate antibiotics. J Bacteriol 190:6014–6025
Fernández E, Weissbach U, Sánchez Reillo C, Braña AF, Méndez C, Rohr J, Salas JA (1998) Identification of two genes from Streptomyces argillaceus encoding glycosyltransferases involved in transfer of a disaccharide during biosynthesis of the antitumor drug mithramycin. J Bacteriol 180:4929–4937
Gomez-Escribano JP, Bibb MJ (2011) Engineering Streptomyces coelicolor for heterologous expression of secondary metabolite gene clusters. Microb Biotechnol 4:207–215
Hopwood DA (2006) Soil to genomics: the Streptomyces chromosome. Annu Rev Genet 40:1–23
Ingram C, Brawner M, Youngman P, Westpheling J (1989) XylE functions as an efficient reporter gene in Streptomyces spp.: use for the study of galP1, a catabolite-controlled promoter. J Bacteriol 171:6617–6624
Kato J, Funa N, Watanabe H, Ohnishi Y, Horinouchi S (2007) Biosynthesis of γ-butyrolactone autoregulators that switch on secondary metabolism and morphological development in Streptomyces. Proc Natl Acad Sci U S A 104:2378–2383
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical Streptomyces genetics. The John Innes Foundation, Norwich, UK
Lambalot RH, Gehring AM, Flugel RS, Zuber P, LaCelle M, Marahiel MA, Reid R, Khosla C, Walsh CT (1996) A new enzyme superfamily—the phosphopantetheinyl transferases. Chem Biol 3:923–936
Li C, Evans RM (1997) Ligation independent cloning irrespective of restriction site compatibility. Nucleic Acids Res 25:4165–4166
Li J, Guo Z, Huang W, Meng X, Ai G, Tang G, Chen Y (2013) Mining of a streptothricin gene cluster from Streptomyces sp. TP-A0356 genome via heterologous expression. Sci China Life Sci 56:619–627
Li J, Xie Z, Wang M, Ai G, Chen Y (2014) Identification and analysis of the paulomycin biosynthetic gene cluster and titer improvement of the paulomycins in Streptomyces paulus NRRL 8115. In submission
Liu G, Tian Y, Yang H, Tan H (2005) A pathway-specific transcriptional regulatory gene for nikkomycin biosynthesis in Streptomyces ansochromogenes that also influences colony development. Mol Microbiol 55(6):1855–1866
Liu G, Chater KF, Chandra G, Niu G, Tan H (2013) Molecular regulation of antibiotic biosynthesis in Streptomyces. Microbiol Mol Biol Rev 77:112–143
Lu Y, Dong X, Liu S, Bie X (2009) Characterization and identification of a novel marine Streptomyces sp. produced antibacterial substance. Mar Biotechnol 11:717–724
Magdevska V, Gaber R, Goranovič D, Kuščer E, Boakes S, Durán Alonso MB, Santamaría RI, Raspor P, Leadlayd PF, Fujs Š, Petković H (2010) Robust reporter system based on chalcone synthase rppA gene from Saccharopolyspora erythraea. J Microbiol Methods 83:111–119
Marie-Pierre D, Mariana GG, Pierre-Étienne J, Nancy C, Carole B, Ryszard B (2009) Cytosine deaminase as a negative selection marker for gene disruption and replacement in the genus Streptomyces and other actinobacteria. Appl Environ Microbiol 75:1211–1214
Marshall VP, Little MS, Johnson LE (1981) A new process and organism for the fermentation production of volonomycin. J Antibiot 34:902–904
Muller M, Auslander S, Auslander D, Kemmer C, Fussenegger M (2012) A novel reporter system for bacterial and mammalian cells based on the non-ribosomal peptide indigoidine. Metab Eng 14:325–335
Myronovskyi M, Welle E, Fedorenko V, Luzhetskyy A (2011) Beta-glucuronidase as a sensitive and versatile reporter in actinomycetes. Appl Environ Microbiol 77:5370–5383
Ou X, Zhang B, Zhang L, Dong K, Liu C, Zhao G, Ding X (2008) SarA influences the sporulation and secondary metabolism in Streptomyces coelicolor M145. Acta Biochim Biophys Sin 40:877–882
Owen JG, Copp JN, Ackerley DF (2011) Rapid and flexible biochemical assays for evaluating 4′-phosphopantetheinyl transferase activity. Biochem J 436:709–717
Owen JG, Robins KJ, Parachin NS, Ackerley DF (2012) A functional screen for recovery of 4′-phosphopantetheinyl transferase and associated natural product biosynthesis genes from metagenome libraries. Environ Microbiol 14:1198–1209
Paget MS, Hintermann G, Smith CP (1994) Construction and application of streptomycete promoter probe vectors which employ the Streptomyces glaucescens tyrosinase-encoding gene as reporter. Gene 146:105–110
Ralph K, Vartul S, Nicholas PT, Jolanta ZC, Katarzyna W, Paul RH, Chun JC, Govind C, Ahmed HF, Michael G, Paul AH (2012) Draft genome sequence of the human pathogen Streptomyces somaliensis, a significant cause of actinomycetoma. J Bacteriol 194:3544–3545
Reverchon S, Rouanet C, Expert D, Nasser W (2002) Characterization of indigoidine biosynthetic genes in Erwinia chrysanthemi and role of this blue pigment in pathogenicity. J Bacteriol 184:654–665
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning, Cold spring harbor laboratory press. Cold spring harbor, New York
Shirling ET, Gottlieb D (1966) Method for characterization of Streptomyces species. Int J Syst Bacteriol 16:313–340
Susan HF, Celia JB, Keith FC (1987) The glucose kinase gene of Streptomyces coelicolor and its use in selecting spontaneous deletions for desired regions of the genome. Mol Gen Genet 206:35–44
Takahashi H, Kumagai T, Kitani K, Mori M, Matoba Y, Sugiyama M (2007) Cloning and characterization of a Streptomyces single module type non-ribosomal peptide synthetase catalyzing a blue pigment synthesis. J Biol Chem 282:9073–9081
Thomas JH, Richard HB (1997) Use of rpsL for dominance selection and gene replacement in Streptomyces roseosporus. J Bacteriol 179:180–186
Walsh CT, Amy MG, Paul HW, Luis EQ, Roger SF (1997) Post-translational modification of polyketide and nonribosomal peptide synthases. Curr Opin Chem Biol 1:309–315
Wietzorrek A, Bibb M (1997) A novel family of proteins that regulates antibiotic production in streptomycetes appears to contain an OmpR-like DNA-binding fold. Mol Microbiol 25:1177–1184
Yang N (2014) The study of bacteria diversity from the south China sea and Xinjiang desert and chemical investigation of one Streptomyces strain. Student PhD thesis: University of Chinese Academy of Sciences (China)
Yu D, Xu F, Jonathan V, Wang S, Zhan J (2012) An indigoidine biosynthetic gene cluster from Streptomyces Chromofuscus ATCC 49982 contains an unusual IndB homologue. J Ind Microbiol Biotechnol 40:159–168
Yuriy R, Elke B, Bogdan T, Andriy L (2014) Actinomycetes biosynthetic potential: how to bridge in silico and in vivo? J Ind Microbiol Biotechnol 41:387–402
Zhang H, Zhou W, Zhuang Y, Liang X, Liu T (2013) Draft genome sequence of Streptomyces bottropensis ATCC 25435, a Bottromycin-Producing actinomycete. Genome Announc 1:e00019-13
Zhao Y (2013) Metabolic engineering of Streptomyces clavuligerus to improve clavulanic acid production. Student PhD thesis: University of Chinese Academy of Sciences (China)
Acknowledgments
We thank Dr. Mervyn J. Bibb, the John Innes Center, Norwich, UK, for providing the S. coelicolor M1146 strain; Dr. Zhaoxin Lu, Nanjing Agriculture University, Nanjing, China, for providing the Streptomyces sp. GB-2 strain; and Dr. Gongli Tang, Shanghai Institute of Organic Chemistry, CAS, Shanghai, China, for providing the plasmid pTGE33. This work was supported in part by the MOST of China (2013CB734003 and 2015CB150602). L.Z. is an awardee for the National Distinguished Young Scholar Program in China. Y.C. is an awardee for the “Hundred Talents Program” of CAS.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 57 kb)
Rights and permissions
About this article
Cite this article
Li, P., Li, J., Guo, Z. et al. An efficient blue-white screening based gene inactivation system for Streptomyces . Appl Microbiol Biotechnol 99, 1923–1933 (2015). https://doi.org/10.1007/s00253-014-6369-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-6369-0