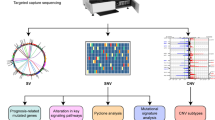
Abstract
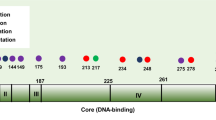
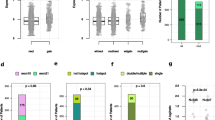
Our recent study showed critical roles of Dmp1 as a sensor of oncogenic Ras, HER2/neu signaling and activation of the Arf-p53 pathway. To elucidate the role of human DMP1 (hDMP1) in breast cancer, one hundred and ten pairs of human breast cancer specimen were studied for the alterations of the hDMP1-ARF-Hdm2-p53 pathway with follow up of clinical outcomes. Loss of heterozygosity (LOH) of the hDMP1 locus was found in 42% of human breast carcinomas, while that of INK4a/ARF and p53 were found in 20 and 34%, respectively. Hdm2 amplification was found in 13% of the same sample, which was found independently of LOH for hDMP1. Conversely, LOH for hDMP1 was found in mutually exclusive fashion with that of INK4a/ARF and p53, and was associated with low Ki67 index and diploid karyotype. Consistently, LOH for hDMP1 was associated with luminal A category and longer relapse-free survival, while that of p53 was associated with non-luminal A and shorter survival. Thus, loss of hDMP1 could define a new disease category associated with prognosis of breast cancer patients. Human breast epithelial cells/cancer cells with wild-type p53 were sensitive to growth inhibition by activated Dmp1:ER while those that delete p14ARF or p53, and/or Hdm2 amplification showed partial or nearly complete resistance, indicating that p53 is a critical target for hDMP1 to exhibit its biological activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hortbagyi GN . Treatment of breast cancer. N Engl J Med 1998; 339: 974–984 Review.
Ross JS, Hortobagyi GN (eds). Molecular Oncology of Breast Cancer. Jones and Bartlett Publishers, Sudbury, Massachusetts, 2005.
Siegel R, Ward E, Brawley O, Jemal A . Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 2011; 61: 212–236.
Chin K, DeVries S, Fridlyand J, Spellman PT, Roydasgupta R, Kuo WL et al. Genomic and transcriptional aberrations linked to breast cancer pathophysiologies. Cancer Cell 2006; 10: 529–541.
Smeets SJ, Harjes U, van Wieringen WN, Sie D, Brakenhoff RH, Meijer GA et al. To DNA or not to DNA/ That is the Question, When It Comes to Molecular Subtyping for the Clinic!. Clin Cancer Res 2011; 17: 4959–4964.
Hirai H, Sherr CJ . Interaction of D-type cyclins with a novel myb-like transcription factor, DMP1. Mol Cell Biol 1996; 16: 6457–6467.
Inoue K, Sherr CJ . Gene expression and cell cycle arrest mediated by transcription factor DMP1 is antagonized by D-type cyclins through a cyclin-dependent-kinase-independent mechanism. Mol Cell Biol 1998; 18: 1590–1600.
Inoue K, Roussel MF, Sherr CJ . Induction of ARF tumor suppressor gene expression and cell cycle arrest by transcription factor DMP1. Proc Natl Acad Sci USA 1999; 96: 3993–3998.
Inoue K, Wen R, Rehg JE, Adachi M, Cleveland JL, Roussel MF et al. Disruption of the ARF transcriptional activator DMP1 facilitates cell immortalization, Ras transformation, and tumorigenesis. Genes Dev 2000; 14: 1797–1809.
Inoue K, Zindy F, Randle DH, Rehg JE, Sherr CJ . Dmp1 is haplo-insufficient for tumor suppression and modifies the frequencies of Arf and p53 mutations in Myc-induced lymphomas. Genes Dev 2001; 15: 2934–2939.
Sreeramaneni R, Chaudhry A, McMahon M, Sherr CJ, Inoue K . Ras-Raf-Arf signaling critically depends on the Dmp1 transcription factor. Mol Cell Biol 2005; 25: 220–232.
Mallakin A, Sugiyama T, Taneja P, Matise LA, Frazier DP, Choudhary M et al. Mutually exclusive inactivation of DMP1 and ARF/p53 in lung cancer. Cancer Cell 2007; 12: 381–394.
Inoue K, Mallakin A, Frazier DP . Dmp1 and tumor suppression. Oncogene 2007; 26: 4329–4335 Review.
Inoue K, Sugiyama T, Taneja P, Morgan RL, Frazier DP . Emerging roles of DMP1 in lung cancer. Cancer Res 2008; 68: 4487–4490 Review.
Sugiyama T, Frazier DP, Taneja P, Morgan RL, Willingham MC, Inoue K . Role of DMP1 and its future in lung cancer diagnostics. Expert Rev Mol Diagn 2008; 8: 435–447.
Taneja P, Maglic D, Kai F, Sugiyama T, Kendig RD, Frazier DP et al. Critical role of Dmp1 in HER2/neu-p53 signaling and breast carcinogenesis. Cancer Res 2010; 70: 9084–9094.
Bieche I, Champeme MH, Matifas F, Hacene K, Callahan R, Lindereau R . Loss of heterozygosity on chromosome 7q and aggressive primary breast cancer. Lancet 1992; 339: 139–143.
Kristjansson AK, Eiriksdottir G, Ragnarsson G, Sigurdsson A, Gudmundsson J, Barkardottir RB et al. Loss of heterozygosity at chromosome 7q in human breast cancer: association with clinical variables. Anticancer Res 1997; 17: 93–98.
Bodner SM, Naeve CW, Rakestraw KM, Jones BG, Valentine VA, Valentine MB et al. Cloning and chromosomal localization of the gene encoding human cyclin D-binding Myb-like protein (hDMP1). Gene 1999; 229: 223–228.
Marchetti A, Buttitta F, Girlando S, Dalla Palma P, Pellegrini S, Fina P et al. Mdm2 gene alterations and mdm2 protein expression in breast carcinomas. J Pathol 1995; 175: 31–38.
Turbin DA, Cheang MC, Bajdik CD, Gelmon KA, Yorida E, De Luca A et al. MDM2 protein expression is a negative prognostic marker in breast carcinoma. Mod Pathol 2006; 19: 69–74.
Masood S . Prognostic/predictive factors in breast cancer. Clin Lab Med 2005; 25: 809–825 viii. Review.
Taneja P, Maglic D, Kai F, Zhu S, Kendig RD, Fry EA et al. Classical and novel molecular prognostic markers for human breast cancer and their clinical significance. Clinical Medicine Insights: Oncology 2010; 4: 15–34.
Gasco M, Yulug IG, Crook T . TP53 mutations in familial breast cancer: functional aspects. Hum Mutat 2003; 21: 301–306.
Wasielewski M, Elstrodt F, Klijn JG, Berns EM, Schutte M . Thirteen new p53 gene mutants identified among 41 human breast cancer cell lines. Breast Cancer Res Treat 2006; 99: 97–101.
Li B, Rosen JM, McMenamin-Balano J, Muller WJ, Perkins AS . neu/ERBB2 cooperates with p53-172H during mammary tumorigenesis in transgenic mice. Mol Cell Biol 1997; 17: 3155–3163.
Cheang MC, Chia SK, Voduc D, Gao D, Leung S, Snider J et al. Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst 2009; 101: 736–750.
Mallakin A, Taneja P, Matise LA, Willingham MC, Inoue K . Expression of Dmp1 in specific differentiated, nonproliferating cells and its repression by E2Fs. Oncogene 2006; 25: 7703–7713.
Mallakin A, Sugiyama T, Kai F, Taneja P, Kendig RD, Frazier DP et al. The Arf-inducing transcription factor Dmp1 encodes transcriptional activator of Amphiregulin, Thrombospondin-1, JunB and Egr1. Int J Cancer 2010; 126: 1403–1416.
Frazier DP, Kendig RD, Kai F, Maglic D, Sugiyama T, Morgan RL et al. Dmp1 physically interacts with p53 and positively regulates p53’s stabilization, nuclear localization, and function. Cancer Res 2012; 72: 1740–1750.
Tschan MP, Fischer KM, Fung VS, Pirnia F, Borner MM, Fey MF et al. Alternative splicing of the human cyclin D-binding Myb-like protein (hDMP1) yields a truncated protein isoform that alters macrophage differentiation patterns. J Biol Chem 2003; 278: 42750–42760.
Ross JS, Linette GP, Stec J, Ross MS, Anwar S, Boguniewicz ADNA . ploidy and cell cycle analysis in breast cancer. Am J Clin Pathol 2003; 120 (Suppl): S72–S84 Review.
Wiesner FG, Magener A, Fasching PA, Wesse J, Bani MR, Rauh C et al. Ki-67 as a prognostic molecular marker in routine clinical use in breast cancer patients. Breast 2009; 18: 135–141.
Shetty A, Loddo M, Fanshawe T, Prevost AT, Sainsbury R, Williams GH et al. DNA replication licensing and cell cycle kinetics of normal and neoplastic breast. Br J Cancer 2005; 93: 1295–1300.
Venkatachalam S, Shi YP, Jones SN, Vogel H, Bradley A, Pinkel D et al. Retention of wild-type p53 in tumors from p53 heterozygous mice: reduction of p53 dosage can promote cancer formation. EMBO J 1998; 17: 4657–4667.
Brosh R, Rotter V . When mutants gain new powers: news from the mutant p53 field. Nat Rev Cancer 2009; 9: 701–713.
Manfredi JJ . The Mdm2-p53 relationship evolves: Mdm2 swings both ways as an oncogene and a tumor suppressor. Genes Dev 2010; 24: 1580–1589 . Review.
Acknowledgements
We are grateful to R Weinberg for HMEC cells; C Sherr, and M Roussel for plasmid DNAs. We thank G Hawkins and Abdoulaye Diallo for LOH analyses of breast cancer samples. We also thank G Kucera for providing information on human breast cancer patients’ samples, Guangchao Sui, Tim Kute for helpful discussions, Guorui Deng, and Jordan Chapman for technical assistance. K Inoue is supported by ACS RSG-07-207-01-MGO, NIH/NCI 5R01CA106314, and by Director’s Challenge Award #20595 from WFUHS. P Taneja was supported by the Susan G Komen Foundation postdoctoral fellowship KG080179. D Maglic has been supported by DOD pre-doctoral fellowship BC100907.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Maglic, D., Zhu, S., Fry, E. et al. Prognostic value of the hDMP1-ARF-Hdm2-p53 pathway in breast cancer. Oncogene 32, 4120–4129 (2013). https://doi.org/10.1038/onc.2012.423
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.423